- Accueil
- h2o
- H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs
H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs
4.8 (581) · € 32.50 · En Stock
A Lewis Structure is a simplified representation of the valence shell electrons in a molecule. Water is made up of two hydrogen atoms and one oxygen atom. Dihydrogen monoxide is another chemical name for this molecule.

Answered: b. Identify the molecular geometry…
Is H2O polar, nonpolar, or ionic? - Quora

Shapes of Molecules

How Water's Properties Are Encoded in Its Molecular Structure and Energies
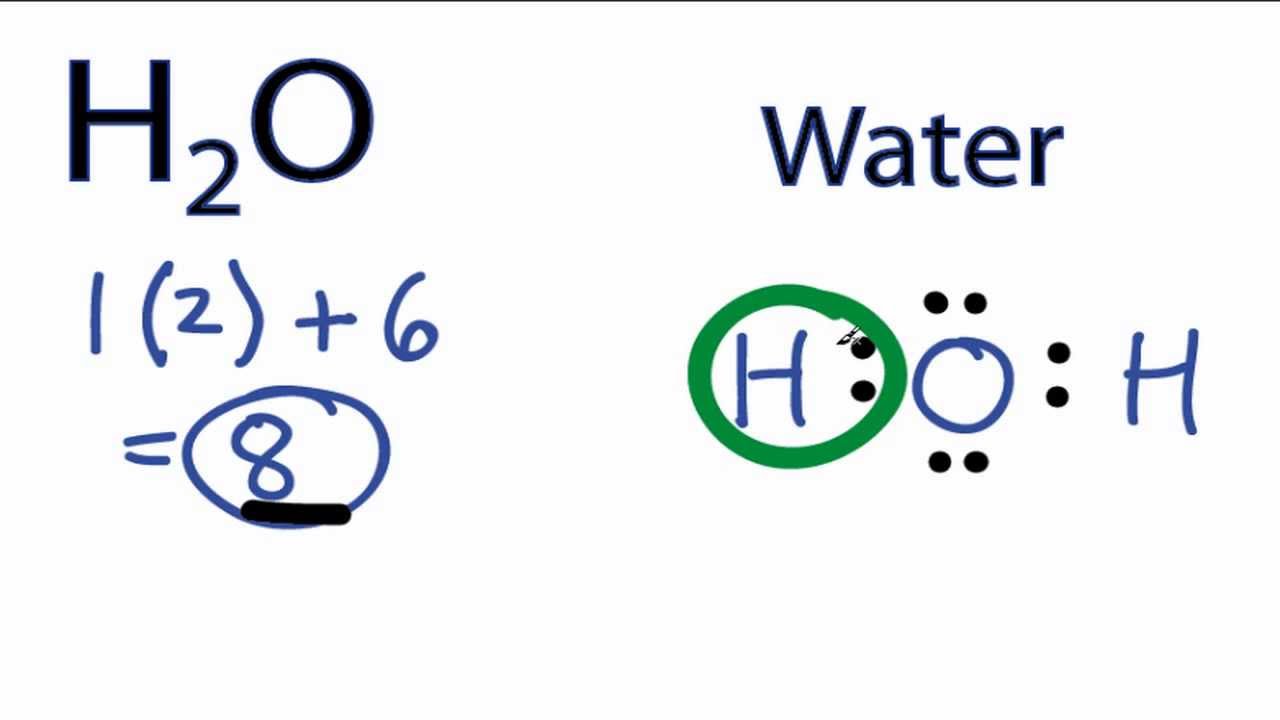
Water Lewis Structure - How to Draw the Lewis Structure for Water

Bond Angles for H2O (Ideal and Actual)

Lewis structure of HCl - How to draw Lewis structure of HCl, Polarity, Molecular Geometry and Hybridization along with FAQs

Water (H2O) and carbon dioxide (CO2) both have one central atom with two atoms bonded to it. However, one is a polar molecule and one is not. Draw the Lewis structure for

Lewis structure of H2O
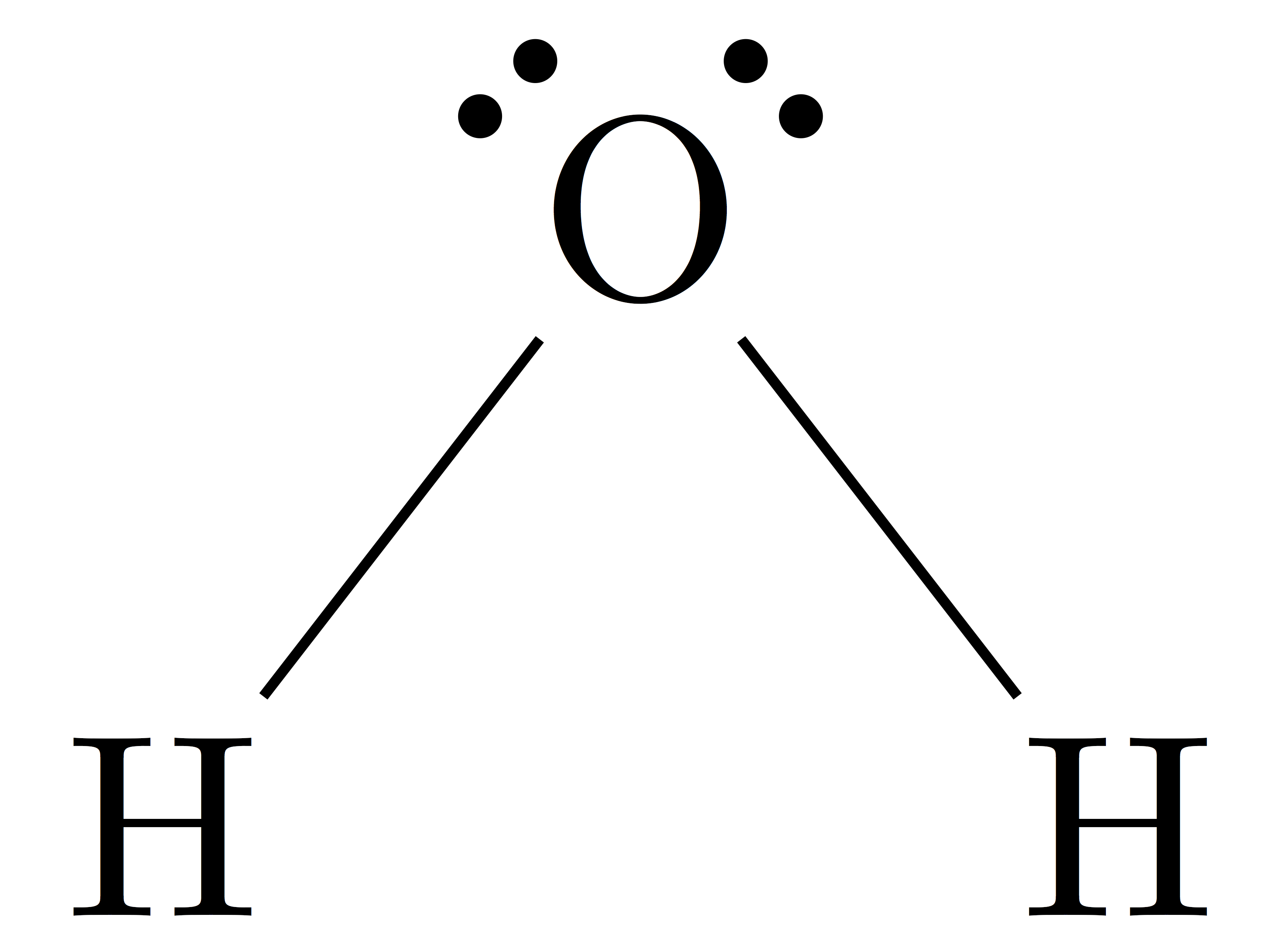
What is the molecular geometry of H_2O? Draw its VSEPR structure.
What if the shape of the water molecule wasn't v-shaped, if the two lone pair electrons stayed on two sides of the oxygen instead of being on the same side? If that

Draw the Lewis structures the following molecules and ions : displaystyle H_{2}S, SiCl_{4}, BeF_{2}, CO^{2-}_{3} and ;HCOOH

Education Sciences, Free Full-Text

H2O Molecular Geometry / Shape and Bond Angle (precise angle is 104.45)